#Keynote 042 trial#
The trial was stopped early due to clear benefit from pembrolizumab over chemotherapy. Responses to pembrolizumab were durable (median duration of response was not reached) which is dramatically different from response to chemotherapy (median DOS of 6.3 months). Flat dosing of pembrolizumab at 200 mg every 3 weeks was shown to have similar benefit compared to patients in KEYNOTE-010 who received 10 mg/kg weight-based dosing. Adverse events were more common in the chemotherapy group, and severe immmune-mediated toxicities were rare. 6.0 months HR 0.50 P<0.001), and median OS was not reached but was also significantly improved with pembrolizumab (HR 0.69 P=0.005). The primary endpoint was progression-free survival (PFS), which was significantly longer in the pembrolizumab group (median PFS 10.3 months vs. In this trial, 305 patients were randomized 1:1 to receive pembrolizumab 200 mg IV every 3 weeks or standard platinum-doublet chemotherapy. This enriched patient population was based upon data from KEYNOTE-001 and KEYNOTE-010 establishing PD-L1 as a positive predictive biomarker for responsiveness to PD-L1 inhibitors. The KEYNOTE-024 trial studied patients with advanced NSCLC and high PD-L1 expression defined as ≥50% of tumor cells (seen in about one quarter of advanced NSCLC patients) and no sensitizing EGFR mutations or ALK translocations.

Patients with tumors with PD-L1 expression ≥50% did even better with pembrolizumab at 2 mg/kg (median OS 14.9 months vs 8.2 months HR 0.54 P=0.0002) and 10 mg/kg (median OS 17.3 months vs 8.2 months HR 0.50 P<0.0001).

KEYNOTE-010 showed that pembrolizumab in patients with previously treated advanced NSCLC with at least 1% PD-L1 expression was superior to docetaxel at both 2 mg/kg (median OS 10.4 vs. Nivolumab was shown to improve overall survival (OS) compared to second-line docetaxel in CheckMate 057, a randomized phase 3 study of 292 patients with advanced nonsquamous NSCLC who progressed during or after platinum-based doublet chemotherapy (median OS 12.2 vs. Further studies demonstrated that targeting the programmed death 1 (PD-1) and programmed death ligand 1 (PD-L1) axis using fully humanized IgG4 monoclonal antibodies against PD-1 (pembrolizumab, nivolumab) or PD-L1 (atezolizumab) was more effective than chemotherapy in first-line metastatic melanoma, second-line advanced renal cell carcinoma, second-line advanced nonsquamous NSCLC, recurrent or metastatic squamous cell carcinoma of the head and neck, and had significant responses in second-line advanced urothelial carcinoma and refractory classical Hodgkin lymphoma. The first approved checkpoint inhibitor was ipilimumab, a monoclonal antibody targeting cytotoxic T-lymphocyte antigen 4 (CTLA-4), in the treatment of melanoma. These agents improve the ability of the immune system to recognize and kill tumor cells. Recent years have seen the approval of immune checkpoint inhibitors for the treatment of multiple malignancies.
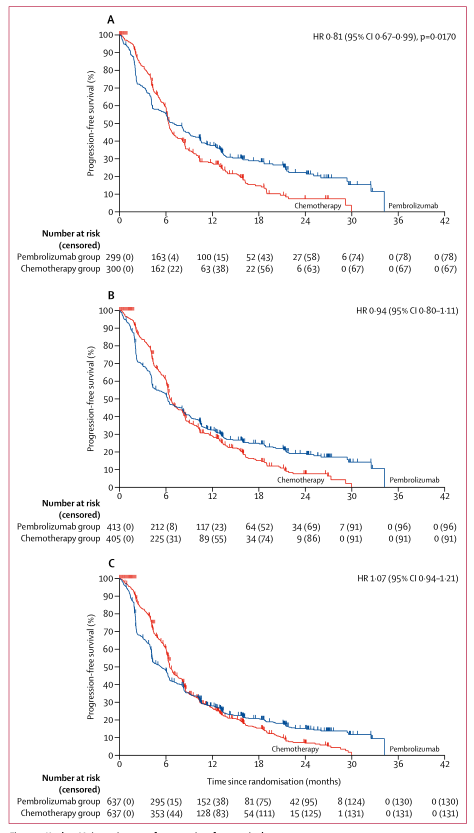
Patients with untreated advanced NSCLC with PD-L1 expression ≥50% had longer progression-free and overall survival with pembrolizumab over platinum-based chemotherapy with fewer adverse events. Among patients with untreated advanced non-small-cell lung cancer (NSCLC) with PD-L1 expression and without EGFR mutations or ALK rearrangements, does pembrolizumab improve progression-free survival compared to platinum-based chemotherapy?
